Toxicology & Cleaning Validation
Scientific exposure limits supporting cleaning validation, toxicological safety and global regulatory compliance.
Indivirtus delivers end-to-end pharmaceutical consulting and CRO solutions—from toxicological risk assessments and regulatory compliance to medical device registration and GMP approvals. Our expertise spans computer system validation, equipment and HVAC calibration, NABL-accredited calibration (thermal, electro-technical, mechanical, pressure, mass & volume), and turnkey greenfield/brownfield projects. We also offer specialized training in GxP, cleaning validation, and quality systems—helping you accelerate approvals, ensure compliance, and scale with confidence.
PDE/OEL Reports as per
EMA | PIC/S | APIC | WHO
Mutagenic & Genotox Assessments
as per ICH, M7

Toxicology & Cleaning Validation
Scientific exposure limits supporting cleaning validation, toxicological safety and global regulatory compliance.

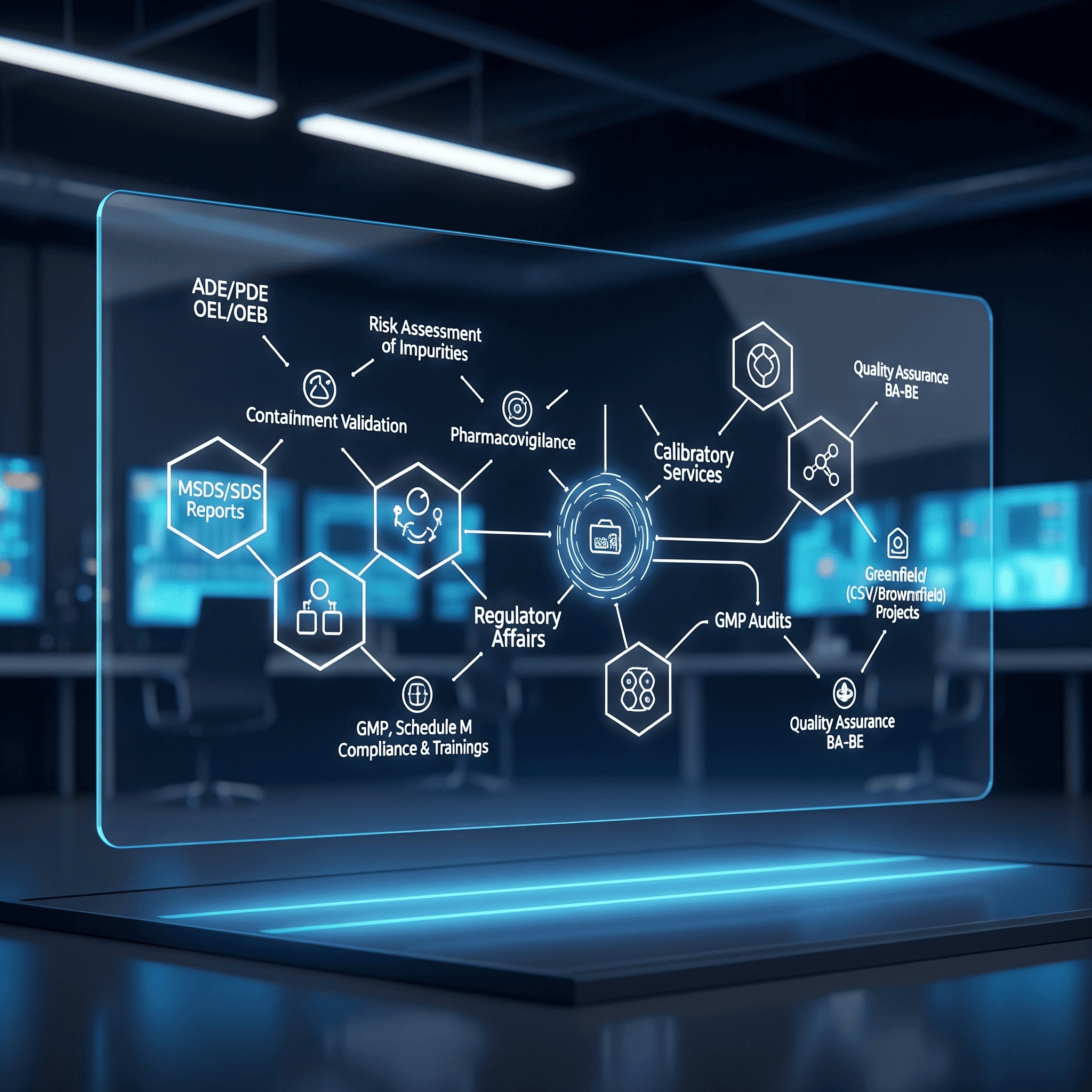
CRO Risk Assessment
Evidence-based impurity evaluation and toxicological risk assessments aligned with global standards.

Containment Validation
SMEPAC-driven containment verification for highly potent compounds and critical manufacturing environments.

Safety Documentation
Compliant MSDS/SDS development for pharmaceutical compounds and safe global handling.

Pharmacovigilance
End-to-end PV support including signal detection, reporting systems and regulatory safety oversight.

Calibration Services
NABL-accredited calibration services maintaining precision, quality systems and audit readiness.

Formulation Development
From pre-formulation research to commercial manufacturing, we help develop robust, effective, and scalable pharmaceutical formulations.

Analytical Development
From analytical method development to validation and regulatory support, we deliver reliable testing solutions for pharmaceutical quality and compliance.

Regulatory Affairs
Technical and strategic support for dossiers, registrations, remediation and global approvals.

Training & Capability
Expert-led capability building across GxP, quality systems, data integrity and regulatory readiness.

GMP Approval Support
Comprehensive support for facility approvals, gap assessments and international regulatory inspections.

Greenfield / Brownfield
Single-window consulting for compliant pharma facilities from concept to qualification.

Validation Services
Comprehensive validation support for automated systems, instruments and critical environments.

Consumer Product Compliance
Regulatory assessments supporting food, cosmetic and consumer products market access.

Independent QA Oversight
QA governance for BA/BE, pre-clinical and clinical studies with regulator-ready compliance.
Deep understanding of USFDA, EMA, CDSCO regulations.
NABL certified labs, GMP-compliant facilities.
Complete solutions from preclinical to tech transfer.
Customized, transparent and responsive engagement.
Qualified experts in science, QA, and compliance.
21 CFR Part 11, GLP/GMP practices ensured.
Join over 1,100+ satisfied clients worldwide
As per our experience in industry your service is one of the best, well trained people.
Industry Professional
Technically sound and professional services. In time delivery of reports at reasonable cost and complying global standards.
Professional Services
Always timely support of documents with high quality assessment through knowledgeable SMEs including reasonable price.
SME / Quality Assessment
Your commitment to delivering quality documents and exceptional service has been instrumental in our success. Their products consistently exceed our specifications.
Quality Documentation Client
This company helped us a lot with providing PDE reports of good quality. I am satisfied about their low pricing and promptness in resolving any issues.
PDE Quality Client